| Protein Purification | BioPatent Communications Contact Me for Discussions About Arts, Technology, and Culture (Blog Me/Pod Me). |
The biotechnology industry is based on purification of genetically engineered proteins for vaccines, anti-cancer drugs, hormones, blood proteins, natural pesticides ... any valuable protein. Genetic engineers isolate the desired DNA sequence and express the protein in a host cell. It is the job of a protein chemist to purify the protein from all the others in the host cell. The diversity of life is based on a diversity of proteins. A protein chemist separates proteins by taking advantage of the differences between proteins.
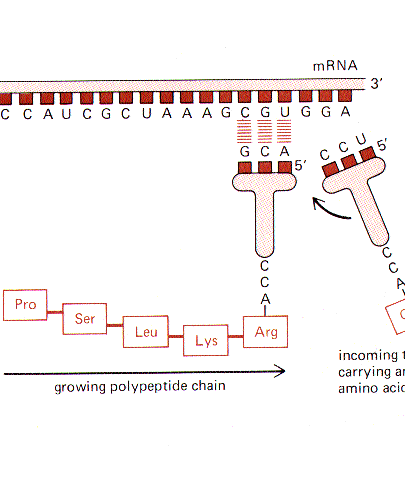 Living cells contain DNA sequences
that are translated into amino acid sequences in a cell to make all the proteins necessary
to life. For example, it takes expression of more than 100,000 different proteins to
make a human being. Proteins are long chains made up of 20 different amino acid
links. Each of the 20 possible amino acid links has a unique combination of electric
charge, fatty character and size. Proteins are all different because of the infinite
number of possible combinations of different amino acid links in each protein chain.
This is what makes albumin in egg whites different from keratin in your hair.
Living cells contain DNA sequences
that are translated into amino acid sequences in a cell to make all the proteins necessary
to life. For example, it takes expression of more than 100,000 different proteins to
make a human being. Proteins are long chains made up of 20 different amino acid
links. Each of the 20 possible amino acid links has a unique combination of electric
charge, fatty character and size. Proteins are all different because of the infinite
number of possible combinations of different amino acid links in each protein chain.
This is what makes albumin in egg whites different from keratin in your hair.
Protein purification requires separation of one desired protein from all the other proteins translated in a cell. This sounds difficult but is easy when you know the characteristics of the protein. The different combinations of amino acids give each protein a unique combination of electric charges, chain lengths, lipid (fatty) character, hydrophilic (water loving) character and shape. A typical protein purification scheme takes advantage of this unique combination of characteristics to separate the desired protein from all the others.
Let's purify an imaginary Protein X that has been expressed in a host cell. Protein X is a large fatty protein with a strong positive charge. Purification starts with disruption of the host cell to release all the internal proteins. Next, proteins in solution are pumped past special resin beads in a cylindrical column. Some proteins will stick to the resin and others will pass through the resin bed. Captured proteins can be released from the resin with special elution solutions.
Cell Extraction. The host cells are grown in a large vat like yeast are grown to make wine or beer. The cells were spun out of the fermentation broth using a centrifuge to create a thick cell paste like peanut butter. The protein chemist resuspends the cell paste in water with detergent. The cell suspension is mixed with small glass beads in a grinding mill to break open the cells. With the cell proteins now in solution, the glass beads and cell debris are easy to remove by centrifugation or filtration.
Hydrophobic Interaction Chromatography (HIC). Hydrophobic proteins tend to stick to other water hating molecules like grease sticks to a plastic bowl in the dirty dishes. Hydrophobic proteins tend to be repelled by salts and water. This is the basis of HIC. HIC resin is like small grains of sand covered with a layer of fat. Hydrophobic proteins, like Protein X, stick when the cell proteins in the water solution are pumped through the HIC resin. Most of the other cell proteins flow straight through to waste.
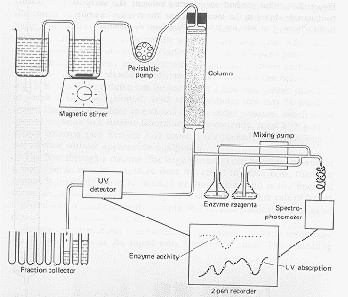 The
Protein chemist adds salt to the water solution of cell proteins to make hydrophobic
proteins stick stronger to the HIC resin. The salty cell extract solution of cell proteins
is pumped through a bed of HIC resin where hydrophobic proteins, including Protein X, get
stuck. Other proteins are washed out with more salt water. Finally, water
without salt, or even water with alcohol or detergent, is pumped through the HIC resin to
release the bound hydrophobic proteins in a HIC elution pool.
The
Protein chemist adds salt to the water solution of cell proteins to make hydrophobic
proteins stick stronger to the HIC resin. The salty cell extract solution of cell proteins
is pumped through a bed of HIC resin where hydrophobic proteins, including Protein X, get
stuck. Other proteins are washed out with more salt water. Finally, water
without salt, or even water with alcohol or detergent, is pumped through the HIC resin to
release the bound hydrophobic proteins in a HIC elution pool.
Ion Exchange Chromatography (IEC). Some amino acid links in a protein chain are negatively charged and some are positively charged. The overall charge of a protein depends on the relative amounts of charged amino acids in the protein chain. IEC resin is like small grains of sand covered with positive (+) or negative (-) charges. Like charges repel and opposite charges attract. Some of the hydrophobic proteins in the HIC elution pool have a negative charge, some have no charge and some, like Protein X, have a positive charge. When the HIC elution pool is pumped through a negatively charged IEC resin, the positively charged proteins stick while other proteins flow through to waste. After the IEC resin is washed, water with strong positively charged salt ions is pumped through the IEC resin to release the proteins. The positively charged salt binds stronger than the positively charged proteins to the negative charges on the IEC resin causing the proteins to fall off in the IEC elution pool (competative elution).
Size Exclusion Chromatography (SEC). Protein X has been followed by some contaminant hydrophobic proteins with positive charges through the purification process so far. However, the contaminant proteins are all sizes depending on the length and shape of their amino acid chains. SEC resin beads contain pores and channels of a certain size. As IEC elution pool is pumped through a tall column of SEC resin, the smaller contaminant proteins spend part of the time loitering inside the SEC beads. The larger Protein X can't fit through the pores and is excluded from the inside of the beads. Protein X flows straight through the SEC column while the smaller proteins, side tracked in the beads, leave the column later. Purified Protein X is collected in the early elution flow before the smaller contaminants have traveled the length of the SEC column.
Protein X was purified by taking advantage of the strong hydrophobicity, strong positive charge and large size. To purify proteins with other characteristics the Protein chemist could: 1) save the proteins that don't stick to the resins, 2) use a HIC resin with more or less fatty character, 3) add more or less salt to the HIC load solution to repel hydrophobic proteins more or less from the solution on to the HIC resin, 4) change the strength or sign (+ or -) of the charges on the IEC resin, 5) change the size of SEC resin pores, 6) collect earlier or later eluting proteins from the SEC column, 7) use other methods such as affinity chromatography where antibodies or plant lectins are attached to resins for very specific binding to desired proteins of very specific shape. The protein chemist can use a broad range of tools and narrowly optimized binding and elution conditions to develop purification processes for any protein.
A process to produce a patentable recombinant protein is typically deemed patentable by law. Other processes must be shown to be new and not obvious compared to older processes in order to be granted patent protection.
Contact Gary at: Baker@BioPatent.com
Return to BioPatent Home: Return
Other BioPatent Pages:
|Patent and Trade
Secret Page| |240Z Renewal Page| |Air-conditioning Repair|
|Performance 240Z| |Lost Wax Casting Page| |240Z Engine Rebuild| |Trademarks|